Co-owner of a General Practice, Chair of Comprehensive Care PHO, member of the General Practice NZ Executive
MB BS 1994 Lond; MRCGP 1998; FRNZCGP 2005

Seven weeks ago, my life turned upside down when I was diagnosed with triple-negative breast cancer.
I am a 50-year-old wife, a mother of three wonderful children, a daughter of very healthy mid 70s parents, a sister, an aunty, a friend, and a dog owner. I also work full time across various roles; a GP, a co-owner and director of a general practice, the Chair of Comprehensive Care PHO in Auckland, and a member of the General Practice New Zealand Executive.
I lead a healthy lifestyle, I have never smoked, drink very little, have amazing cholesterol and sugar levels, keep fit and active and have had an annual mammogram for the last decade.
I want to share with you my story to enhance awareness of triple negative breast cancer and the limited treatment options available.
My diagnosis came completely out of the blue. One day I was fighting fit and well and the next I had an aggressive breast cancer. My world changed overnight. I went from healer to patient, mother and housewife to invalid, carer to cared for.
As well as being a working GP, I have spent the last year and a half completely invested in managing COVID in our community, both at my practice level and at PHO level in the Waitemata area. Setting up swabbing clinics, PPE supplies, vaccinations for front line staff and then campaigning to get vaccinations into primary care. My other focus and passion has been on driving equity in health outcomes, developing a new strategy for our PHO to ensure that Māori are given care in novel ways that entitles them to the same health expectations as Pakeha.
I remember the sense of utter elation I felt on the eve of my 50th birthday, back in March, when I received my first Pfizer vaccine. Like all health professionals, I had been terrified of contracting COVID through my work and my greatest fear was either giving it to my family or dying and leaving my children motherless. It was such a relief to me to feel protected and bulletproof. Now, I am immunosuppressed and vulnerable to COVID once again.
Ironically, it was COVID and the start of the lockdown that led to my breast cancer diagnosis. Routine staff temperature checks identified that I had a fever which I was completely unaware of. I went through the process of investigation, nothing obvious came up, and eventually I realised it could be an unusual presentation of a malignancy, a paraneoplastic syndrome. I managed to rebook my COVID cancelled mammogram and bingo, a 2.6cm breast tumour.
I was lucky to be able to get a biopsy done immediately after the mammogram. Five days later, in mid-September, I saw a breast surgeon for the results.
That was the most distressing day of my life. The results were given to me in a very honest and direct way. The surgeon told me the words I never wanted to hear, “You have breast cancer. We are going to manage it very aggressively to try to treat it.” I stopped processing anything after the word “try”. I held my husband’s hand, and just focused on breathing, feeling utter terror. I would like to say I took the news in a composed and dignified way, but my shock was just too great to respond, or to cry. I just completely shut down, trying to disassociate from what was happening around me.
My breast surgeon told me I did not have time to wait in public for chemotherapy, as I had such aggressive breast cancer. She also told me I needed to stop work immediately.
Seven weeks on, my sleep is still haunted and disturbed by that day and the days that followed. My grief and horror were not for myself, they were for the shocking impact I knew my diagnosis was going to have on my husband, my children, and my family. I was powerless to protect them. Telling my children and parents was the worst thing I have ever had to do in my life. At least I could tell my 14-year-old daughter face to face and hug her as she sobbed. I had to find the courage to tell my sons over Zoom because one was at university in Christchurch, and the other was in Wellington. Neither could return to a locked down Auckland.
Now for the good news. I count my many blessings that within a week I had a portacath put in, genetic testing, a clip put in my tumour, saw an oncologist, had a PET CT scan and started chemotherapy. I was very lucky the cancer was localised and had not yet spread to my lymph nodes or beyond. They had caught it early. I am very fortuitous to have health insurance to cover some of the costs of going privately. My heart bleeds for those that do not, who find themselves in my shoes and who must wait for the mercy of the public system to coordinate all of this. I am told of long waits in public for chemotherapy.
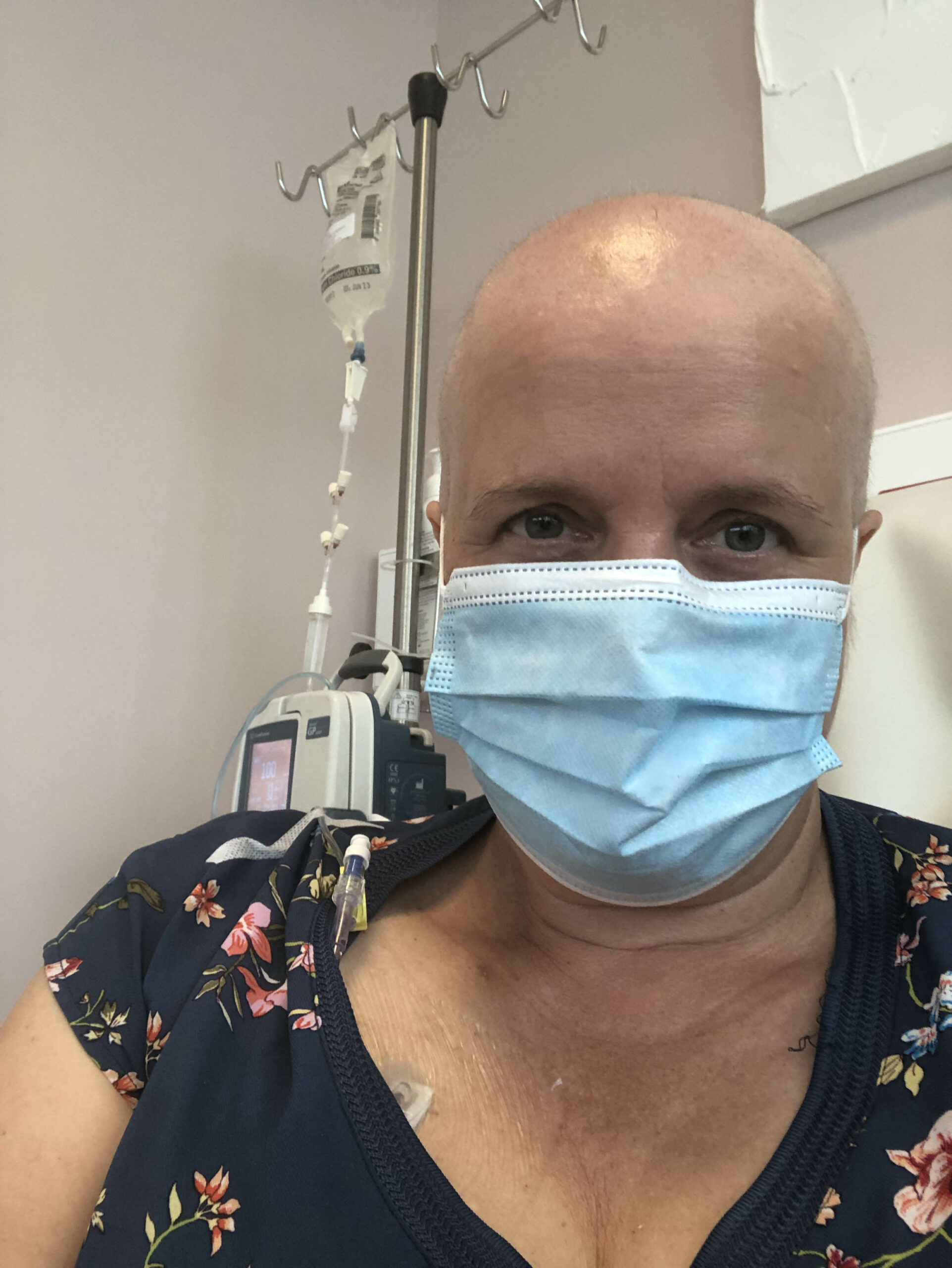
I am also extremely lucky to have a wonderful oncologist who bravely confronted the difficult truth; the treatment options funded in New Zealand are obsolete and if I wanted gold standard treatment to give me the best chance of survival, I would need to fund it myself.
I am seven doses of weekly chemotherapy treatments into a 6-month course. It will be followed by surgery and then a course of radiation. In addition, I am self-funding an immunotherapy, Pembrolizumab (more commonly known as Keytruda) which I will have every 3 weeks for a year. I have lost my hair and I am very tired, but I am handling the treatment well so far. I am not able to work, and I am reliant on the support of my husband, daughter, family and friends.
I realise how privileged I am that my husband and I had savings and can afford to fund the $7,800 every three weeks for Pembrolizumab. Fortunately, the patient cost contribution is capped at $60,000 + GST (approximately $69,000) and the manufacturer, Merck, Sharpe & Dohme, will take over payments for as long as the patient needs it. However, the cost of the administration of the Pembrolizumab remains at the cost of the patient, whether treated in private or public. Please also note that the Government is adding $9,000 of GST to a medicine it won’t fund, making it even more expensive and out of reach to more women.
This treatment, although expensive, is clearly giving me the best possible chance against triple negative breast cancer and will hopefully save my life or extend it greatly. To my family the cost is worth it, and I recognise what a privileged position I am in.
My conscience is struggling to deal with the fact that other women in New Zealand are not being given the same chance at life. I feel guilty, I am no more deserving than anyone else. Our health system, that I have devoted my life to working for, is not moving forward as science produces lifesaving solutions. It breaks my heart when I think of the women who are not even offered or cannot afford life-saving drugs for their triple negative breast cancer. I am ashamed to think that Givealittle has become one of the main funders of modern cancer drugs in New Zealand, for what is standard care internationally. Can you imagine what it must be like having to rely on the charity of others to get the healthcare you urgently need?
I have come to the sad conclusion that women with triple negative breast cancer are betrayed by the public health system in New Zealand. Why do we not count? Do our lives not matter enough? Does our contribution to society mean so little that we are left with ineffective treatment for a disease that will otherwise kill us? It astounds me that our health system is not rushing to fund a treatment for early disease that will be cost effective if it prevents women developing metastatic disease, which is going to be far more expensive to manage. It is short sighted, unfair and is inflicting unnecessary suffering and harm on many women and their whanau.
It is well documented that Māori wahine do far less well with breast cancer compared to Pakeha women. We have a government who say they are focused on getting equity of health outcomes, yet by not funding drugs that make a substantial difference to survival, they are increasing the equity gap further. How come this is not obvious to them?

On the election campaign trail in 2017, the Labour Party promised access to new cancer medicines but has so far failed to deliver. Successive governments have underfunded cancer treatments to the extent that New Zealand is ranked 20th in the OECD.
We are shamefully placed at the bottom of the developed world for per-capita spend on modern medicines. Countries like Mexico, Colombia, Latvia, and Belarus are ahead of us. Between 2011 and 2018 the number of publicly reimbursed modern cancer medicines in New Zealand was 7, out of a total of 90 possibilities. This compares to 35 in Australia and 60 in the UK. Do not believe for a moment it is because these drugs are outrageously expensive to fund. The cost of Pembrolizumab for a year is on par to the cost of a year of renal dialysis, or one coronary artery bypass graft, or two knee replacements. These are all things we expect out health system to fund, so why not modern cancer drugs?
As a GP I have always had respect for my colleagues working at Medsafe and Pharmac, whom I believe do their very best to get New Zealanders the safest drugs at the best price with an impossibly small budget to work with. The problem is a political one, these agencies are chronically underfunded, they just do not have the budget for modern day medicines and are not set up to have the agility to act quickly when new drugs become available. And it is not just cancer drugs, there are many other drugs languishing on the waiting lists for approval.
You may ask why I am writing this Perspective; it is certainly not for sympathy as I consider myself very lucky to be on the best medication possible right now.
I am writing this because I am upset about this injustice in New Zealand. All our people deserve healthcare that meets international standards, and all deserve equity of outcomes, not just the wealthy.
I am writing this to heighten public awareness that our drug funding in New Zealand is abysmal, in the hope that politicians will listen and act fast.
I am writing this for the other women with early or late triple negative breast cancer, in the hope that all of them can get the best treatment possible NOW, before it is too late.

And I am also writing this for the women who believe themselves well today, but are only one diagnosis away from being me.
Dr Heidi MacRae – treating Triple Negative Breast Cancer Most people consider breast cancer to be just one entity, but the reality is there is a huge variation between the different types. Many people have made well-meaning but misinformed comments to me about how treatable breast cancer is nowadays, not realising that triple negative has been one of the exceptions. It only accounts for 15% of breast cancer, typically affects women under 40 and is commonly associated with BRAC1 gene mutations. Triple negative breast cancer was only first described in 2005, over a decade after I finished medical school. Its name refers to the fact that it does not have receptors for oestrogen, progesterone, or human epidermal growth factor receptor 2 (HER2). Those receptors are the mainstay of treatment options for other types of breast cancer. Cancer drugs target the receptors and without them it makes the cancer very difficult to treat, meaning survival rates for triple negative breast cancer have been very poor and recurrence rates high within a few years. My oncologist told me how lucky I was as there were new successful studies that she would base my treatment on. She encouraged me to read them, but in those first dark weeks I could not bear to look, I could not bring myself to read anything that would confirm to me what I remembered about how bad triple negative breast cancer was. My husband, who is not medical, read the science and became very confident that I could beat this. It took me 6 more weeks to bring myself to look at the science to confirm for myself that there was a reason to be positive. The New England Journal of Medicine published the Pembrolizumab (Keytruda) for Early Triple-Negative Breast Cancer trial in Feb 2020 (KEYNOTE-522). Pembrolizumab, an anti–programmed death 1 (PD-1) monoclonal antibody, a type of immunotherapy, had previously been shown to have antitumour activity in patients with metastatic triple-negative breast cancer. This KEYNOTE 522-study looked at using immunotherapy (the Pembrolizumab) in addition to preoperative chemotherapy (called neoadjuvant chemotherapy) in over 1000 women in 21 countries with early triple negative breast cancer, just like mine. 65% of women in the Pembrolizumab plus chemotherapy group, compared to 51% in the placebo–chemotherapy group had a complete pathological response to treatment (meaning when the breast tissue was removed the tumour was completely gone). It was not until 2021 that the follow up results come in looking at survival data. Pembrolizumab plus chemotherapy reduced the risk of death by 27% and the risk of disease progression or death by 34% compared to chemotherapy alone. “Keytruda is the first immunotherapy to show positive results for event-free survival in patients with high-risk early-stage TNBC, a particularly aggressive form of breast cancer,” said Dr. Roy Baynes, chief medical officer, Merck Research Laboratories. “The improvement in pathological complete response rates initially observed following pre-operative treatment was encouraging, and now that we are seeing the data mature after four years to include a statistically significant improvement in event-free survival, we look forward to working with the FDA and other global authorities to bring this new option to patients as quickly as possible. “ Merck were true to their promise and in July 2021, the FDA approved Pembrolizumab for high-risk, early-stage, triple-negative breast cancer in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery. In October 2021 the European Commission followed suit. Clearly the US and EC have drug agencies that are nimble and responsive to new research for conditions that are difficult to treat and have high morbidity and mortality. The disappointing news is that it is unlikely that there will be approval granted in New Zealand for many years if at all. The waiting list to fund prioritised modern medicines (which are MedSafe approved and recommended for funding by Pharmac’s own clinical advisory panel) is growing. The average time an approved medicine remains on the waitlist in New Zealand is 4.7 years. |

Show your support.
Please share Dr Heidi MacRae’s perspective story.
New Zealand needs access now to modern medicines to treat Breast Cancer Triple Negative




Other Voices
Let’s close the Gap
Get Involved1.
REFORM
PHARMAC
Achieve a measurable, political commitment to reform Pharmac and create a fit-for-purpose drug-buying agency that supports and enables greatly improved access to modern medicines – and ensure a direct line of political accountability.
2.
OVERHAUL THE FUNDING
METHODOLOGY
Introduce a globally accepted modern, cost-benefit analysis for medicines and medical devices which looks at the ‘value’ of a medicine, and considers the financial, economic, and social impact of untreated disease on our society.
3.
COMMIT TO AN OUTCOMES-BASED MEDICAL STRATEGY
Develop a Medicines Strategy to guide the decision-making process, create measurable targets to reduce Pharmac’s waiting list, and detail how the agency will respond to rapid developments in modern medicine to improve health outcomes for New Zealanders.